Why Belgium Is Strong in HealthTech and Biotech
March 14, 2026 by Harshit Gupta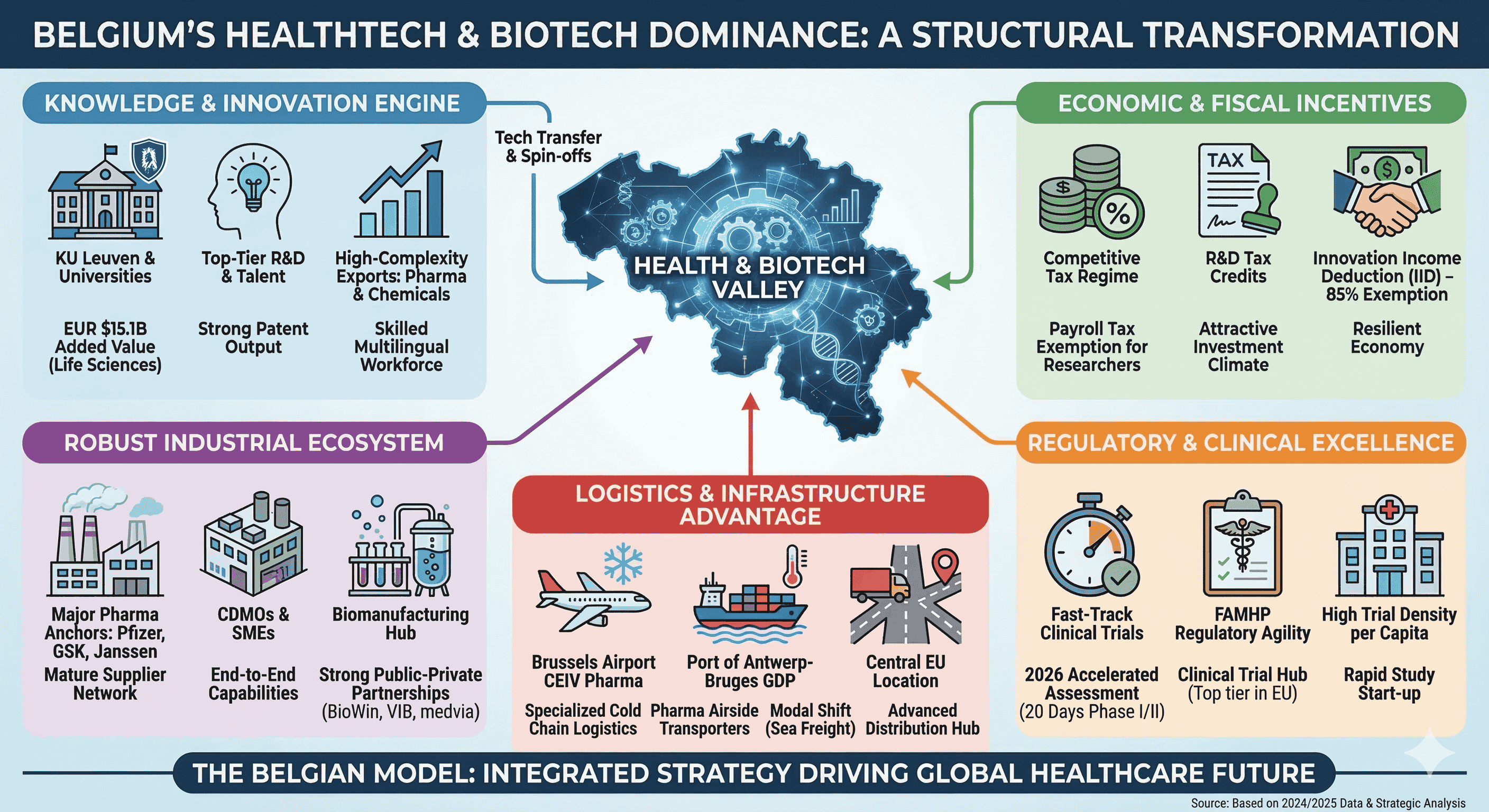
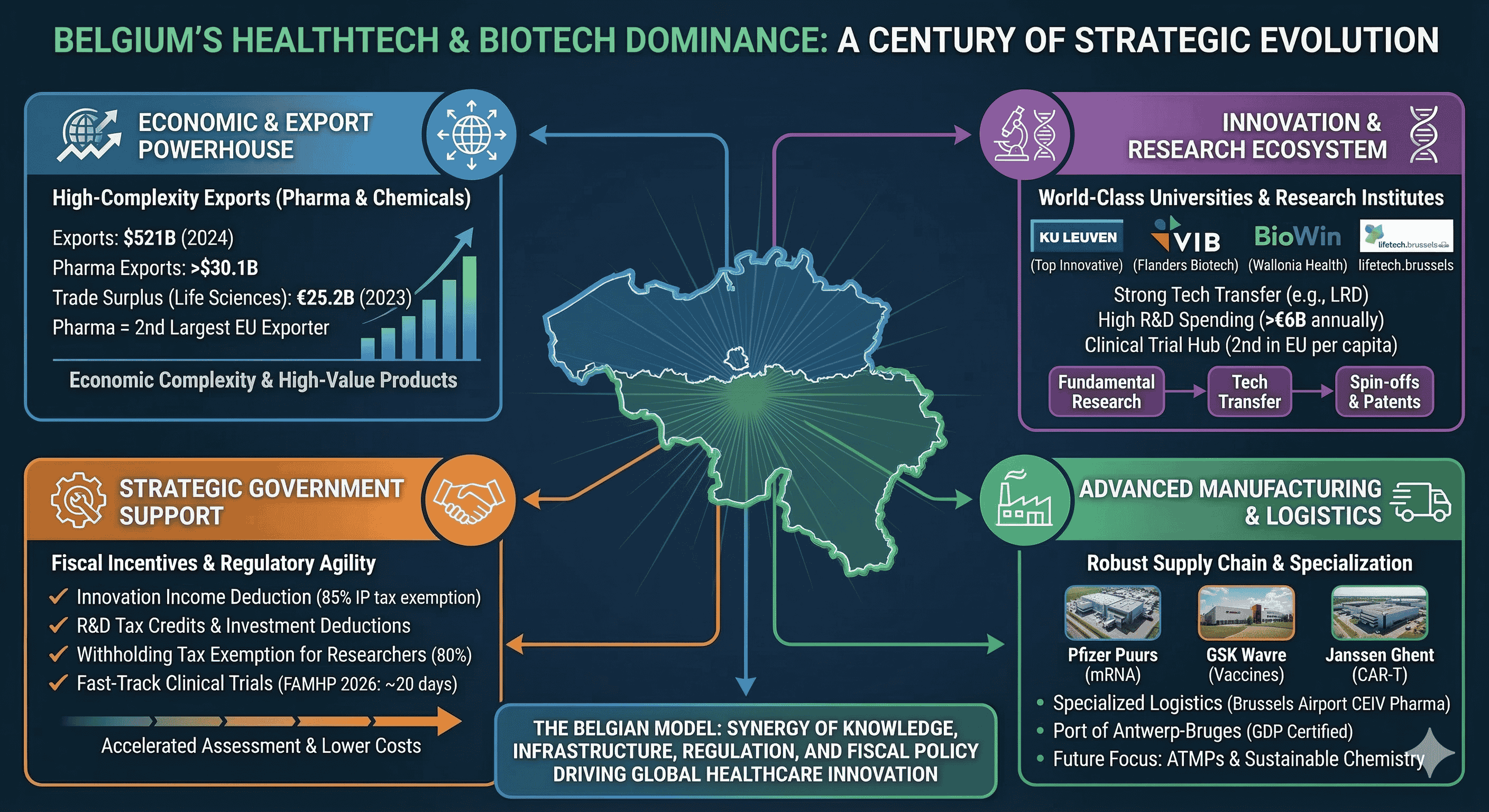
Belgium’s prominence in the global health technology and biotechnology sectors is not a product of serendipity but the result of a deliberate, century-long structural transformation. By 2024, Belgium exported goods valued at USD $521 billion, and while broader export dynamics have faced an average annual decline of 1.5% over the last five years, the biopharmaceutical and chemical segments have emerged as resilient pillars of high complexity. Economic complexity, a metric developed by the Harvard Growth Lab, suggests that countries specializing in products requiring sophisticated, non-transferable knowledge are better positioned for long-term growth. Belgium’s export basket is heavily weighted toward these high-complexity products, particularly pharmaceutical products and mineral fuels, which have expanded their global market share even as traditional machinery manufacturing has receded. This evolution signifies a completed structural transformation where the nation has successfully entered nearly all major high-productivity sectors, anchored by a life sciences ecosystem that generates EUR $15.1 billion in added value and sustains a trade surplus of EUR $25.2 billion.
Macroeconomic Resilience and the Life Sciences Export Engine
The Belgian economy has demonstrated notable resilience in the face of recent global shocks, with real GDP growth stabilizing at approximately 1.0% to 1.2% in 2024 and 2025. While export-oriented industrial sectors broadly contracted, domestic demand remained supported by the automatic indexation of wages, a mechanism that preserved purchasing power but also emphasized the need for high-value-added sectors to offset rising labor costs. In this context, the biopharmaceutical industry acts as a vital stabilizer. In 2024, exports of medicaments reached over USD $30.1 billion, with broader medicinal and pharmaceutical products totaling USD $31.5 billion. This sector is highly concentrated; more than 14% of all European pharmaceutical exports originate in Belgium, making it the second-largest exporter in the European Union.
The reliance on world trade is a defining characteristic of the Belgian model, with exports representing over two-thirds of the country's gross national income. The pharmaceutical sector’s role in this trade profile is disproportionately large relative to the country’s population of approximately 11.5 million. The following table illustrates the trade dynamics and the pivotal role of the pharmaceutical and chemical sectors in the Belgian export economy.
Economic Indicator | 2024/2025 Metric (USD/EUR) | Source |
Nominal GDP (Sep 2025) | USD $188.2 billion | |
GDP Per Capita (Dec 2024) | USD $56,730.7 | |
Total Exports (2024) | USD $521 billion | |
Pharmaceutical Exports (Medicaments) | USD $30.18 billion | |
Chemical Products Exports | EUR $6.52 billion (Monthly Dec 25) | |
Trade Surplus (Chemicals & Life Sciences) | EUR $25.2 billion (2023) |
Belgium's strategic approach to economic growth involves diversifying know-how into "nearby" products in the product space—moving from traditional chemicals into complex biologics, vaccines, and advanced therapy medicinal products (ATMPs). This transition is facilitated by a highly skilled, multilingual workforce and a central geographic location that allows for rapid distribution across the European market.
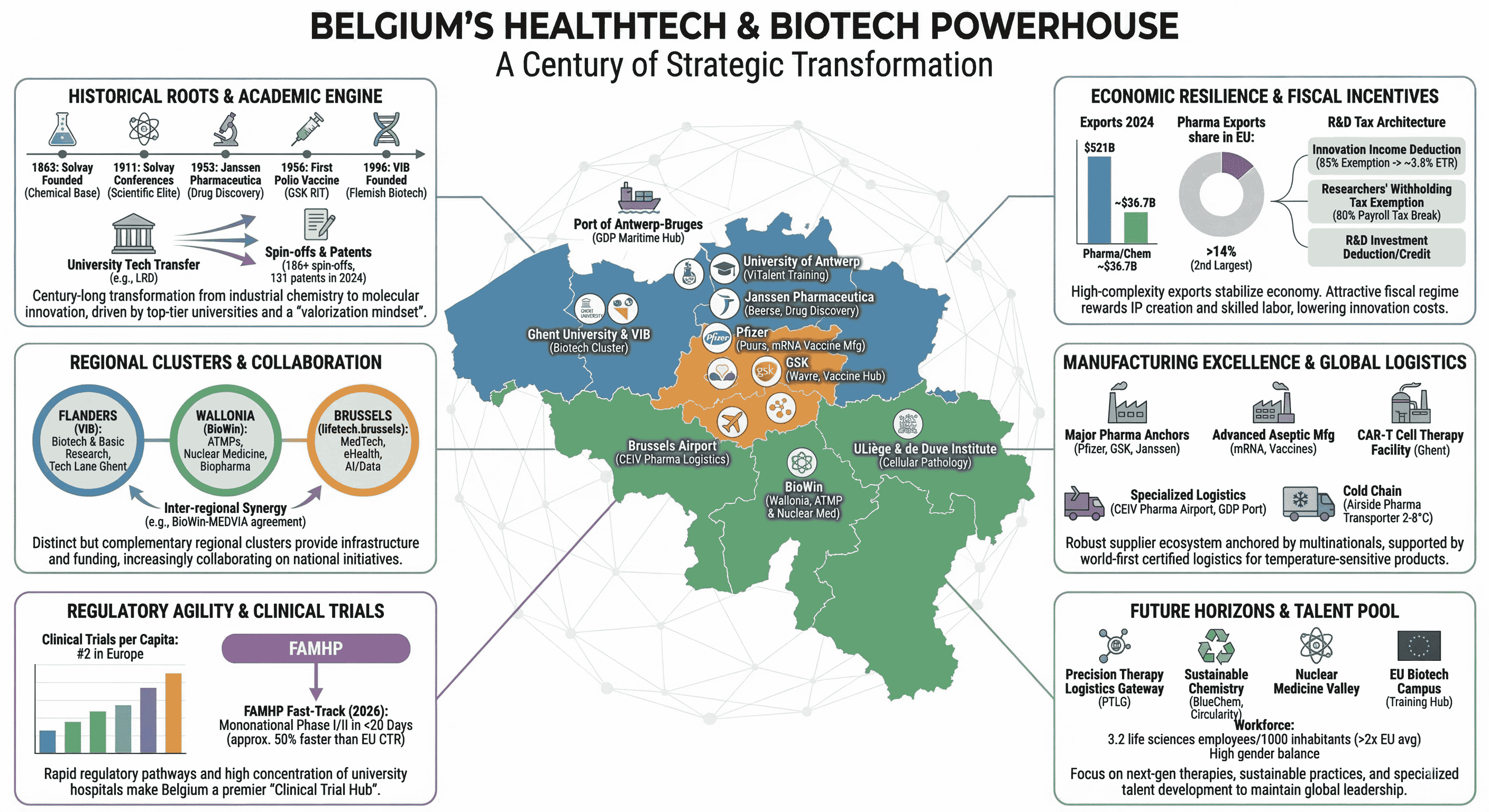
Historical Genesis: From Industrial Chemistry to Molecular Innovation
The roots of Belgium’s leadership in health technology can be traced back to the late 19th and early 20th centuries, when the nation became a global hub for chemical engineering. The founding of Solvay in 1863 by Ernest Solvay created a global chemical empire that not only provided the industrial base for future pharmaceutical manufacturing but also fostered a culture of elite scientific inquiry. The legendary Solvay Conferences, initiated in 1911, gathered the world’s leading physicists and chemists in Brussels, cementing the city’s reputation as a center for scientific progress.
The mid-20th century saw the transition from general industrial chemicals to specialized medicinal research. In 1933, Constant Janssen acquired the rights to distribute Richter pharmaceutical products in the Benelux region, a move that eventually led his son, Dr. Paul Janssen, to establish Janssen Pharmaceutica in 1953. Dr. Paul Janssen, often referred to as a legend in pharmacology, focused on understanding the chemical structure-effect relationship, leading to the development of over 80 medical compounds and the filing of more than 100 patents. The acquisition of Janssen Pharmaceutica by Johnson & Johnson in 1961 provided the Belgian firm with the capital and global reach to expand its research campus in Beerse into one of the world’s most productive drug discovery hubs.
Simultaneously, Belgium became a pioneer in immunology and virology. In 1956, the Research and Industry for Therapeutics (RIT), which would later become the vaccines division of GlaxoSmithKline (GSK), produced the world’s first polio vaccine. This set the stage for Belgium to become a global leader in vaccine production, a position reinforced during the COVID-19 pandemic when the Pfizer facility in Puurs became the primary European site for mRNA vaccine manufacturing.
Milestone Year | Event / Achievement | Impact on Sector |
1863 | Founding of Solvay | Establishment of industrial chemical base |
1911 | First Solvay Conference | Global positioning in fundamental science |
1928 | Founding of UCB | Introduction of innovative chemical/pharma focus |
1953 | Founding of Janssen Pharmaceutica | Birth of independent Belgian medical research |
1956 | First Polio Vaccine (GSK RIT) | Foundation of world-leading vaccine cluster |
1963 | Upjohn (Pfizer) Puurs facility | Start of advanced aseptic manufacturing |
1996 | Founding of VIB | Formalization of the Flemish biotech cluster |
1996 | Listing of Innogenetics | First biotech listing on European exchange |
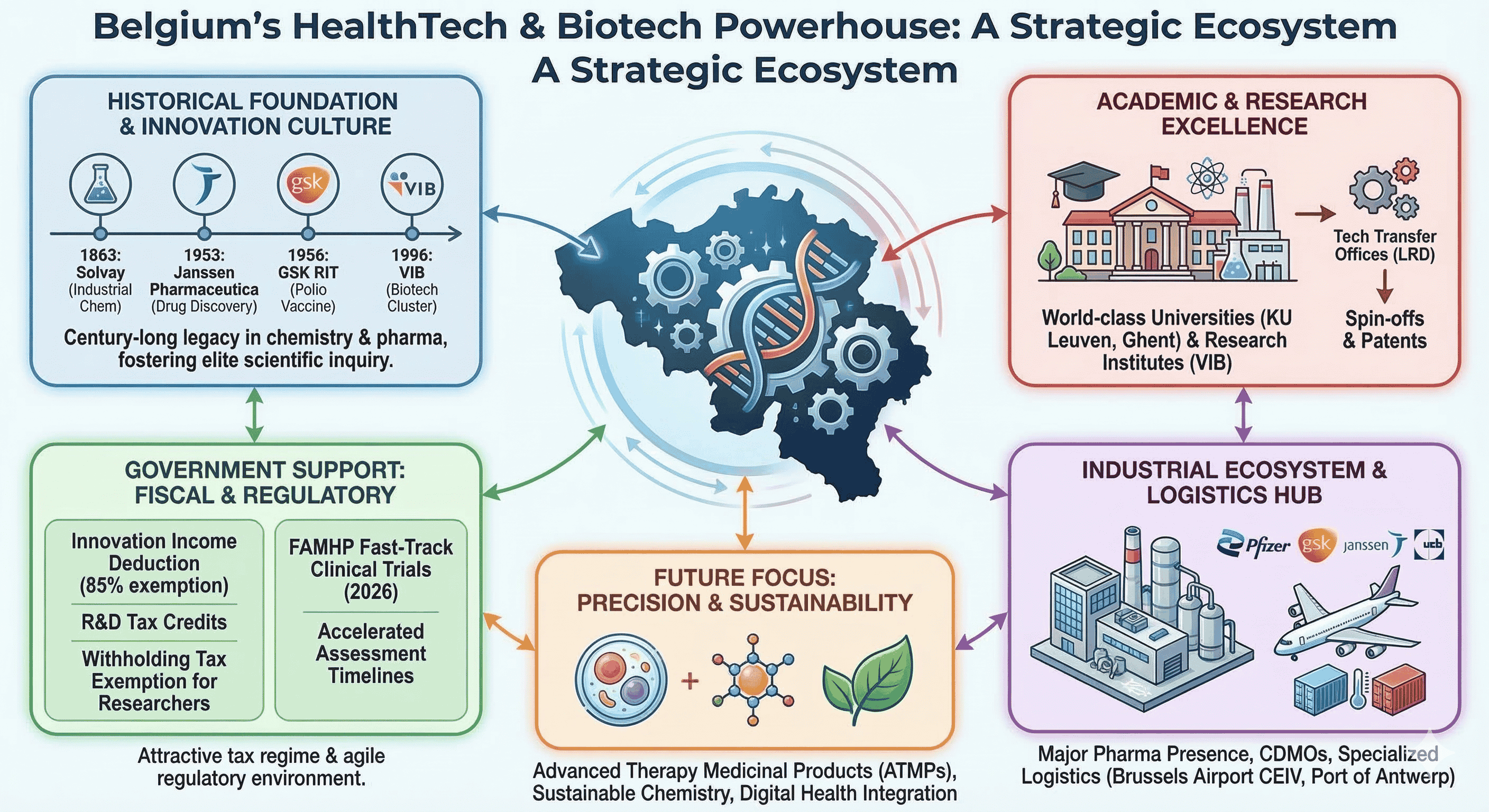
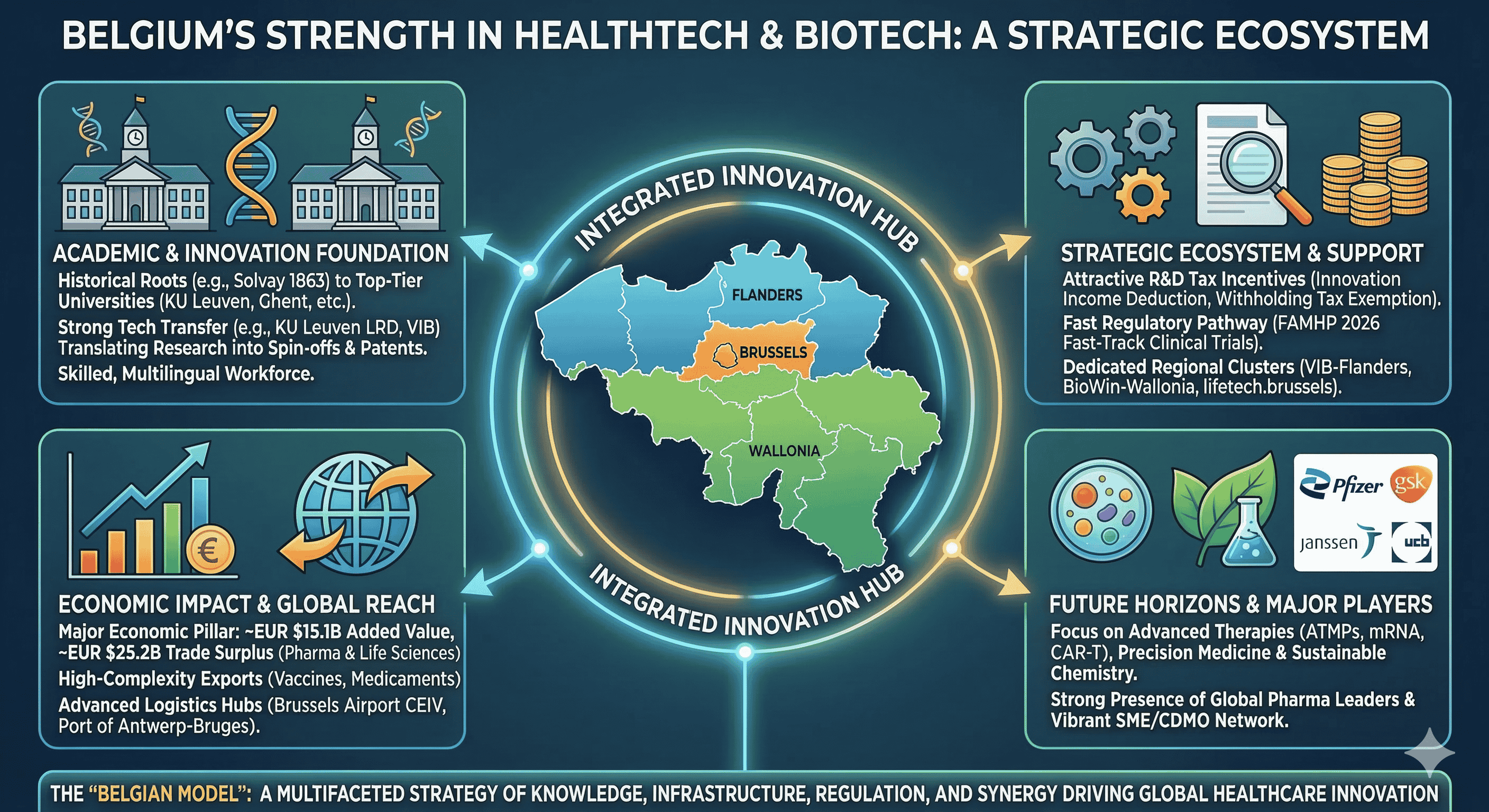
The Academic Foundation and Technology Transfer Mechanisms
The strength of Belgium’s biotech sector is intrinsically linked to its university system, which excels in translating fundamental research into commercial applications. KU Leuven, founded in 1425, consistently ranks among the most innovative universities globally, holding the 7th position in the Reuters ranking and often leading the European list. In 2024, KU Leuven reported a research expenditure of EUR $749.2 million, with 81% of funding coming from external sources, including competitive regional, national, and European grants, as well as industrial contracts.
A critical component of this success is KU Leuven Research & Development (LRD), the university's technology transfer office. Established in the early 1970s, LRD has institutionalized a "valorization mindset" among academics. LRD lowers the threshold for patenting by bearing the initial costs of filing in the priority year and providing specialized IP and patent officers to manage the legal process. This has resulted in a portfolio of 186 spin-offs and 131 new patents in 2024 alone. The university hospital (UZ Leuven) further complements this by providing a clinical environment for testing new therapies.
Beyond KU Leuven, other institutions like Ghent University (ranked 6th in European patent applications among universities) and the University of Antwerp (ranked 10th) contribute significantly to the Flemish biotech cluster. Ghent is particularly strong in agricultural biotechnology (AgBio), while Antwerp hosts specialized training centers like ViTalent. In Wallonia, the de Duve Institute and the University of Liège have pioneered breakthroughs in cellular pathology and oncology, leading to spin-offs such as Mithra Pharmaceuticals.
Regional Cluster Strategy: Flanders, Wallonia, and Brussels
Belgium’s decentralized governance has allowed for the development of distinct but complementary regional life sciences clusters. These clusters serve as catalysts by providing infrastructure, funding, and networking opportunities.
Flanders: VIB and the "Health and Biotech Valley"
The Vlaams Instituut voor Biotechnologie (VIB) is a central pillar of the Flemish ecosystem. As a research institute, VIB focuses on fundamental life sciences, but its mandate includes a strong focus on economic impact. In 2023, VIB contributed nearly EUR $1 billion in Gross Value Added (GVA) to the Flemish economy and supported over 11,000 jobs. For every EUR $1 of core funding from the Flemish government, VIB returns EUR $12 in economic impact. VIB operates several incubators and accelerators, such as the Tech Lane Ghent Science Park, which provides 25,000 m2 of specialized infrastructure for the biotech community.
Wallonia: BioWin and the ATMP Specialization
BioWin, the Walloon healthcare competitiveness cluster, focuses on biopharma, nuclear medicine, and medical technology. Its mission is to drive the growth of the health innovation ecosystem by connecting over 250 member organizations, including SMEs, universities, and hospitals. BioWin is particularly focused on Advanced Therapy Medicinal Products (ATMPs), coordinating projects like the ATMP-PIT, which has a portfolio worth EUR $81.7 million and involves 21 companies. Wallonia also leverages a historical advantage in nuclear medicine, utilizing isotopes for cancer diagnostics and radioligand therapies.
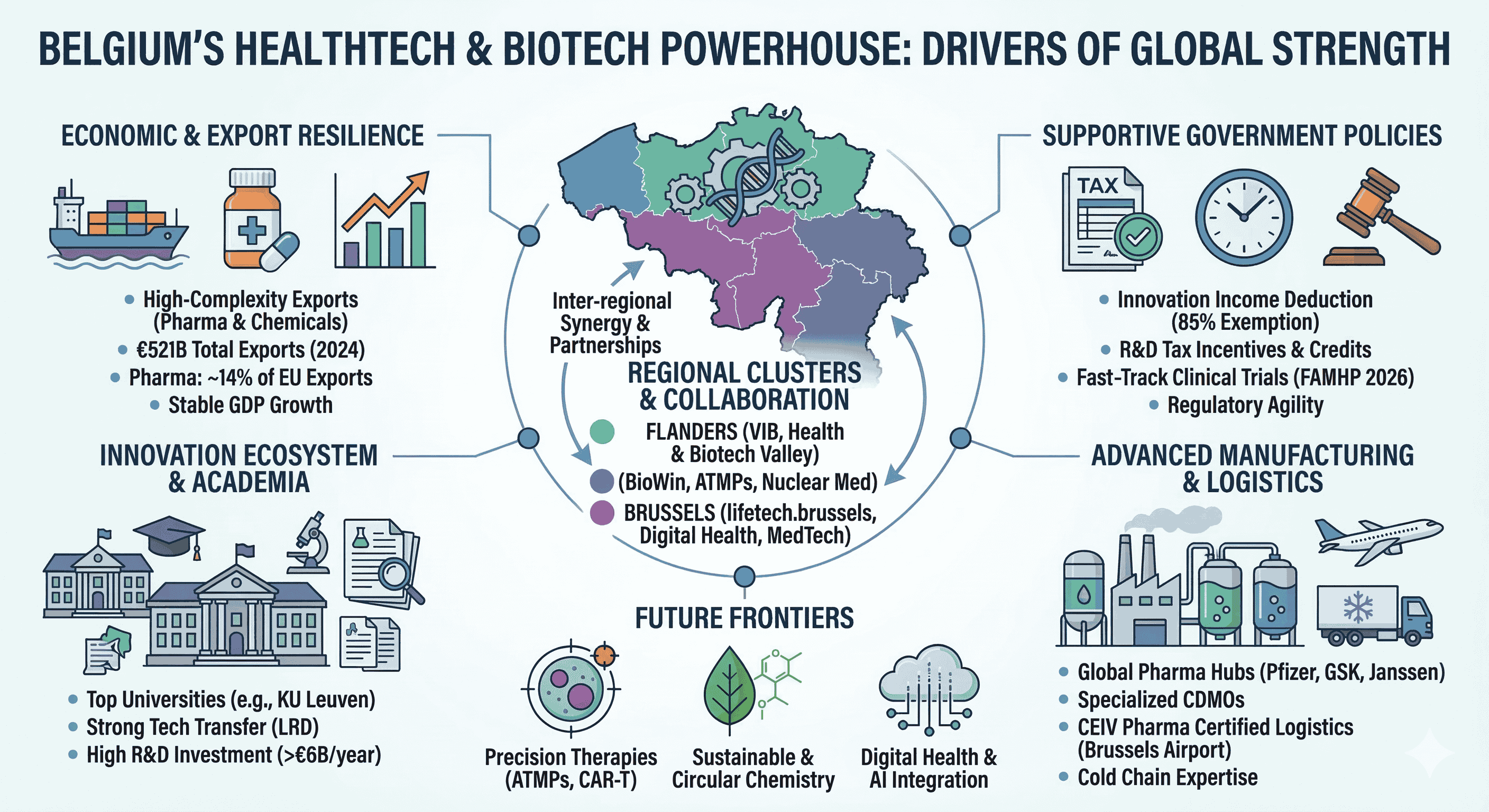
Brussels: lifetech.brussels and Digital Health
The lifetech.brussels cluster serves as the gateway to the Brussels health innovation ecosystem, specializing in MedTech and eHealth. Its MedTech Accelerator program is an intensive six-month initiative designed to boost startups through workshops, coaching, and networking with decision-makers and investors. The cluster also emphasizes the integration of AI and data analytics, preparing members for the challenges and opportunities of the European Health Data Space (EHDS).
Inter-regional Collaboration
While regional identities are strong, the 2023-2024 strategic agreement between BioWin (Wallonia) and MEDVIA (Flanders) marks a significant shift toward national synergy. This partnership aims to harmonize funding flows and launch collaborative projects, particularly in ATMPs and biomanufacturing, to attract foreign investors and maintain Belgium’s international reputation as a leading life sciences cluster.
Fiscal Incentives and the R&D Tax Architecture
Belgium’s fiscal landscape is one of the most attractive globally for R&D-intensive companies. The government provides a comprehensive suite of tax incentives that cover the entire innovation cycle, from initial research costs to the commercialization of intellectual property.
Innovation Income Deduction (IID)
The Innovation Income Deduction is a key Belgian tax measure that provides an 85% corporate income tax exemption for qualifying intellectual property income. This effectively reduces the maximum effective corporate tax rate on IP-derived income to approximately 3.8%. The deduction applies to patents, supplementary protection certificates, and copyright-protected software resulting from R&D. A crucial feature for 2025 is the option for companies to convert unused portions of this deduction into a non-refundable tax credit, which can be carried forward indefinitely—a strategic adaptation to the OECD Pillar Two global minimum tax rules.

Withholding Tax Exemption for Researchers
To mitigate high labor costs, Belgian law allows employers an 80% exemption from the payment of withholding tax on the salaries of researchers. This incentive applies to employees holding PhDs, Master’s, or specific Bachelor’s degrees in STEM fields who are engaged in fundamental research, industrial research, or experimental development. This measure significantly lowers the total cost of innovation and helps Belgian firms compete for global talent.
R&D Investment Deduction and Tax Credit
For investments in R&D equipment or patent acquisitions, companies can choose between a one-time technology deduction or a spread deduction over the depreciation period. For 2025, the one-time deduction stands at 13.5%, while the spread deduction is 20.5%. If a company does not have sufficient taxable profit to utilize the investment deduction, the remaining balance can be converted into a refundable tax credit after four years.
Incentive Type | Benefit / Rate | Eligibility Criteria | Source |
Innovation Income Deduction | 85% tax exemption (3.8% ETR) | Patents, software, orphan drugs | |
Withholding Tax Exemption | 80% exemption on payroll tax | STEM degrees, 80%+ time on R&D | |
Technology Deduction (One-time) | 13.5% of asset value | New R&D equipment, laboratories | |
Technology Deduction (Spread) | 20.5% over depreciation period | New R&D equipment | |
Digital Investment Deduction | 20% one-off deduction | Capitalized digital assets |
Clinical Research Excellence and Regulatory Agility
Belgium’s status as a "Clinical Trial Hub" is a cornerstone of its strength. It consistently ranks as the second country in Europe (after Denmark) in terms of pharmaceutical industry-sponsored clinical trials per capita. In 2024, approximately 2.47 trials were authorized per 100,000 inhabitants, significantly higher than in much larger nations like Germany (rank 2nd in total trials but lower per capita) and France.
The Federal Agency for Medicines and Health Products (FAMHP) plays a critical role in maintaining this competitiveness. From January 1, 2026, the FAMHP introduced accelerated assessment timelines for clinical trial applications, aiming to reduce study start-up times by nearly 50%. These expedited procedures allow mononational Phase I, I/II, and II trials to be assessed within a maximum of 20 days. This regulatory agility ensures that patients in Belgium gain faster access to innovative treatments, particularly in oncology and rare diseases, which together account for over 50% of the country’s trial authorizations.
Trial Phase (Mononational) | 2026 Validation Phase | 2026 Eval. & Decision | Total Fast-Track |
Phase I, I/II, II | 5 Days | 15 Days | 20 Days |
Phase III, IV | 7 Days | 28 Days | 35 Days |
Multinational (Pilot) | 7 Days | 28 Days | 35 Days |
These timelines are approximately half as long as the standard European Clinical Trials Regulation (CTR) maximums, positioning Belgium among the fastest regulators in Europe. The focus on speed is balanced with a rigorous scientific assessment, often supported by the high concentration of university hospitals and clinical experts available in the country.
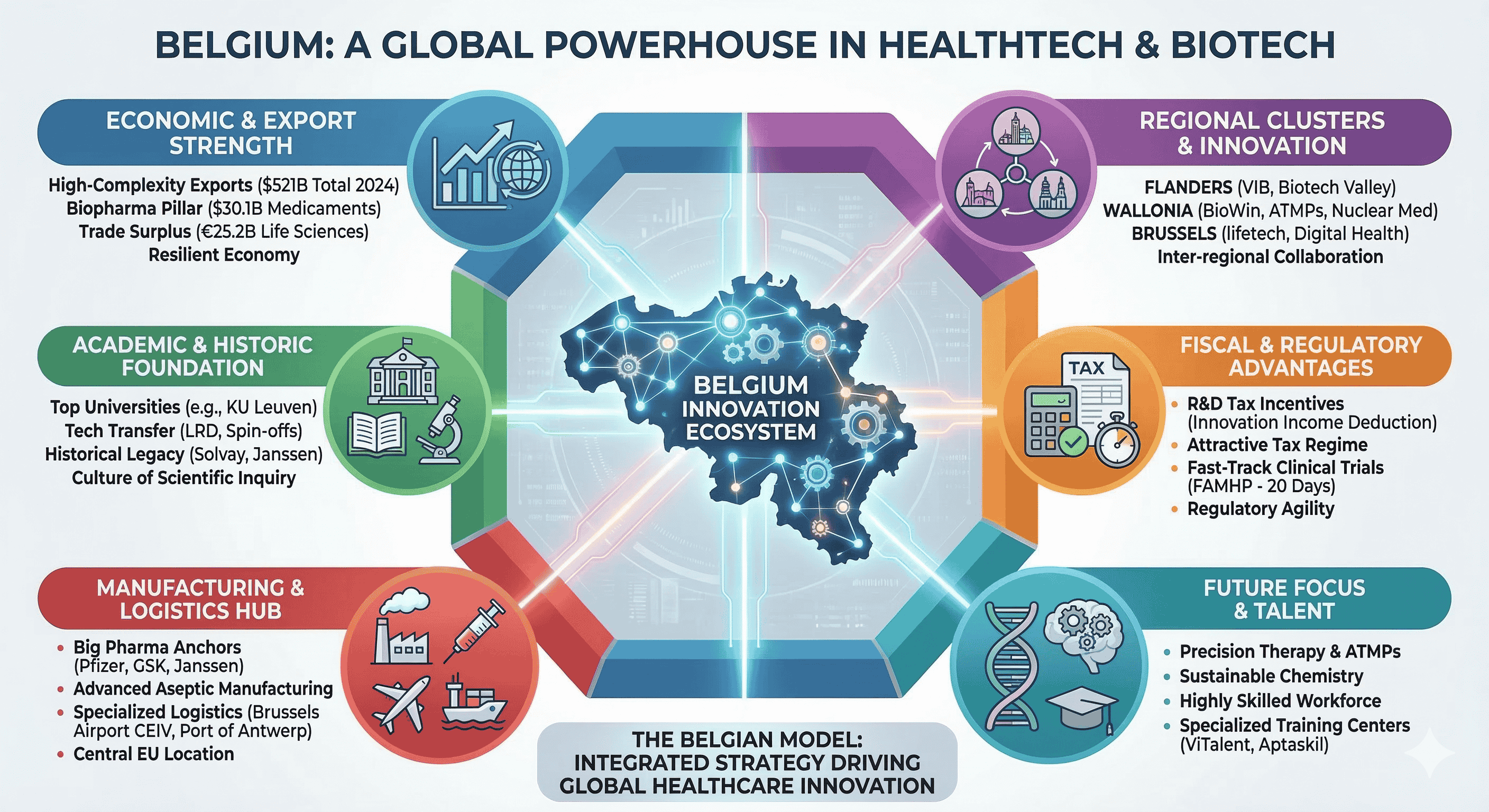
The Manufacturing and Logistics Ecosystem
The "anchoring effect" of large pharmaceutical multinationals has created a robust supplier ecosystem in Belgium. Companies like Pfizer, GSK, Janssen, and UCB have consistently invested in the country, attracting a network of service providers, specialized logistics firms, and contract development and manufacturing organizations (CDMOs).
Pfizer Puurs and the mRNA Revolution
The Pfizer site in Puurs is one of the company’s oldest and most advanced aseptic production facilities in Europe. Established in 1963, it now employs 4,500 people. During the pandemic, it became a central node in the global COVID-19 vaccine network. In December 2022, Pfizer announced an additional EUR $1.2 billion investment to further expand production capacity, cold storage capabilities, and packaging options at the Puurs site, aiming to support a pipeline of 19 new products.
GSK Wavre: Global Vaccine Hub
GSK’s vaccines division is headquartered in Belgium, with its Wavre site serving as the world’s largest vaccine manufacturing facility. GSK employs significant adjuvant technology developed at its Rixensart R&D site to produce one billion doses annually. GSK’s presence has a profound multiplier effect; it partners with local nanotechnology centers like imec to automate biomanufacturing processes and provides a talent pool for the broader ecosystem.
Janssen’s CAR-T Facility in Ghent
Janssen (Johnson & Johnson) employs over 5,000 people in Belgium and maintains end-to-end capabilities from discovery to commercialization. A recent milestone is the selection of Tech Lane Science Park in Ghent for the company’s first European CAR-T cell therapy manufacturing center. This facility uses genetically modified T-cells to treat blood cancer, representing the forefront of precision medicine and leveraging Belgium’s central location for time-sensitive logistics.
Logistical Infrastructure and CEIV Certification
Belgium’s logistics sector is specialized to handle the high standards required for life sciences. Brussels Airport was the first in the world to receive the CEIV Pharma certification, a standard for the transport of products requiring an unbroken cold chain. The airport features 45,000 m2 of temperature-controlled storage and has developed the "Airside Pharma Transporter" to maintain temperature ranges such as 2−8∘C and 15−25∘C on the tarmac.
The Port of Antwerp-Bruges is similarly advanced, being the world’s first maritime port to operate its entire pharmaceutical supply chain according to European Good Distribution Practice (GDP) rules. This allows for a "modal shift," where companies like UCB can transport large volumes of biologics via sea freight—which is cheaper and has a lower carbon footprint—while maintaining strict quality standards.
Workforce Development and Specialized Training
The sustainability of the Belgian biotech sector depends on its ability to produce and retain high-level talent. Belgium has a solid pool of life sciences employees, with 3.2 per 1,000 inhabitants—more than twice the European average. To meet the demand for specialized biomanufacturing and digital skills, several dedicated training centers have been established.
ViTalent (Flanders): Located in Niel, this center serves as a job simulator for the pharma and biotech industry. It features a technical training hall with simulated production lines and cleanrooms to train employees, job seekers, and students in realistic working conditions.
Aptaskil (Wallonia): Based in Seneffe, Aptaskil specializes in production-related jobs. It trains 4,500 people annually and is expanding to Liège to accommodate a goal of 6,000 trainees per year.
EU Biotech Campus (Wallonia): Scheduled to open in 2025 in Charleroi, this campus will act as a business accelerator and training hub, focusing on digital and biomanufacturing skills using innovative methods like VR/AR and digital twins.
Beyond technical skills, the sector demonstrates a commitment to diversity. In 2024, the biopharmaceutical sector achieved a balanced gender distribution with 50% female employees. Notably, women are even better represented in research (62%) and production (67%), and 63% of directors in the sector are women, indicating that the traditional "glass ceiling" is increasingly a thing of the past in Belgian pharma.
Future Horizons: Precision Therapy and Sustainable Chemistry
Belgium is positioning itself to lead in the next generation of healthcare innovations. This includes a focus on precision therapies, sustainable chemistry, and the digitalization of the value chain.
Precision Therapy Logistics Gateway (PTLG)
The PTLG project, launched in early 2025, is a collaboration between Brussels Airport, Pharma.Aero, and the Antwerp ATMP ecosystem. It aims to develop a standardized international protocol for the transport of highly sensitive cell, gene, and radioligand therapies. This ensures that as treatments become personalized, the logistics chain can support the transport of a patient’s own cellular material with extreme speed and accuracy.
Sustainable and Circular Chemistry
The chemical and life sciences sector is undergoing a "double twin transition" toward climate neutrality and digitalization. Initiatives like BlueChem, the first incubator for sustainable chemistry in the Benelux, focus on CO2 utilization, bio-based raw materials, and chemical recycling. This transition is essential for the sustainable anchoring of the industry in Belgium, ensuring that the country remains a leader in "green" biomanufacturing.
Nuclear Medicine and the de Duve Legacy
Building on the legacy of Nobel laureate Christian de Duve, Wallonia is reinforcing its leadership in nuclear medicine. The region’s expertise in medical isotopes and radiopharmaceutical production is being channeled into targeted cancer therapies. The collaboration between BioWin and international partners aims to create a "Nuclear Medicine Valley" that integrates diagnostic and therapeutic capabilities into a single clinical pathway.
Strategic Challenges and Outlook
Despite its strengths, the Belgian health technology sector faces challenges. The high public debt-to-GDP ratio (projected at 110% in 2025) and fiscal consolidation needs may impact public research funding. Additionally, global competition from South-East Asia and the Western Pacific is intensifying, with those regions now surpassing Europe in total clinical trial volume. Belgium's response is to double down on its high-complexity strategy—reinforcing its talent pool, unlocking the potential of health data, and accelerating the adoption of innovative platforms like mRNA and cell therapy.
The transition from "Invented in Belgium" to "Made in Belgium" is a key strategic goal. By strengthening local bioproduction capacity through CDMOs and large-scale manufacturing investments, Belgium aims to ensure that the innovations developed in its world-class university labs are also produced within its borders. This integrated approach—combining fundamental research, a favorable fiscal regime, regulatory speed, and world-class logistics—constitutes the "Belgian Model" that continues to drive its success in HealthTech and biotechnology.
Final Summary of Competitive Determinants
Belgium’s success in life sciences is the result of a multifaceted strategy:
Knowledge Concentration: Sustained by high-complexity exports and a top-tier innovation ranking.
Infrastructure Advantage: Specialized logistics hubs like Brussels Airport and the Port of Antwerp-Bruges.
Regulatory Efficiency: The 2026 FAMHP fast-track system for clinical trials.
Fiscal Competitiveness: A tax regime that rewards IP creation and skilled labor.
Industrial Synergy: A strong "anchoring effect" from global leaders like GSK and Pfizer.
As the sector continues to grow, with R&D investments exceeding EUR $6 billion annually, Belgium remains a primary "Health and Biotech Valley," shaping the future of global healthcare through scientific excellence and operational precision.
The Real Startup Playbook: Ideas, Growth, Funding, and Founder Psychology
Building a startup is exciting, chaotic, and often misunderstood. If you’ve ever wondered how founders actually validate ideas, find co-founders, raise funding, and survive the emotional rollercoaster, these articles explore the realities behind the startup world.
Start with How to Validate Your Startup Idea in 48 Hours for $0 to understand how founders test ideas quickly before wasting months building the wrong product. Once you have an idea, the next challenge is building it—From Idea to MVP: A Step-by-Step Guide for Solo Founder walks through the journey from concept to a real product.
Many founders struggle with partnerships and team decisions. Should your co-founder live in the same city? Explore Remote vs. Local: Does Your Co-Founder Need to Live in the Same City? and learn the risks hidden in partnerships in 5 Red Flags to Look for When Choosing a Startup Partner. If you’re searching for technical help, How to Find a Technical Co-Founder (Without a Six-Figure Salary) explains how founders actually do it.
The startup journey isn’t just about building products—it’s also about mindset and decision-making. Articles like Decision Fatigue: The Silent Startup Killer, Fear vs Logic: How Founders Actually Make Decisions, and How Overthinking Destroys Early Momentum reveal the psychological battles founders face behind the scenes.
Growth is another misunderstood part of startups. Learn why strategy matters more than hype in Ideas Don’t Scale. Systems Do. and why discipline matters in Why Your Startup Doesn’t Need Growth — It Needs Focus. Discover the importance of early traction in How the First 100 Users Decide Your Startup’s Fate and understand team building through The First Hire That Actually Matters.
Funding is often romanticized, but reality is more complex. Why First-Time Founders Should Avoid Big Funding challenges common assumptions about venture capital, while Revenue Solves More Problems Than Funding explains why sustainable businesses matter more than investor money.
The startup ecosystem also varies by geography. If you're curious about India’s startup scene, explore The New Playbook for Raising in Bangalore, Why Raising Pre-Seed in Bangalore Is Harder Than Ever, and Why B2B SaaS from Bangalore Scales Faster.
But startups are not just about strategy and growth—they’re also about people and emotions. Articles like The Hidden Burnout of Bangalore Founders, Comparison Culture in India’s Startup, and The Loneliness of First-Time Founders in Bangalore reveal the personal struggles founders rarely talk about publicly.
And finally, if you want a deeper perspective on the startup journey itself, explore Lessons Learned Too Late by First-Time Founders and The Myth of the “Overnight Success” Startup—because the truth behind startup success is far more complex than it appears.